Energy flows directionally through ecosystems, entering as sunlight (or inorganic molecules for chemoautotrophs) and leaving as heat during energy transformation between trophic levels. Rather than flowing through an ecosystem, the matter that makes up organisms is conserved and recycled. The six most common elements associated with organic molecules—carbon, nitrogen, hydrogen, oxygen, phosphorus, and sulfur—take various chemical forms and may exist for long periods in the atmosphere, on land, in water, or beneath Earth’s surface. Geologic processes, such as weathering, erosion, water drainage, and the subduction of continental plates, all play a role in the cycling of elements on Earth. Because geology and chemistry have major roles in studying these processes, recycling inorganic matter between living organisms and their nonliving environment is called biogeochemical cycles.
Organisms use the six aforementioned elements in a variety of ways. Hydrogen and oxygen are found in water and organic molecules, both essential to life. Carbon is found in all organic molecules, whereas nitrogen is an important component of nucleic acids and proteins. Phosphorus is used to make nucleic acids and the phospholipids that comprise biological membranes. Lastly, sulfur is critical to the three-dimensional shape of proteins.
The cycling of these elements is interconnected. For example, water movement is critical for leaching sulfur and phosphorus into rivers, lakes, and oceans. Minerals cycle through the biosphere between the biotic and abiotic components and from one organism to another.
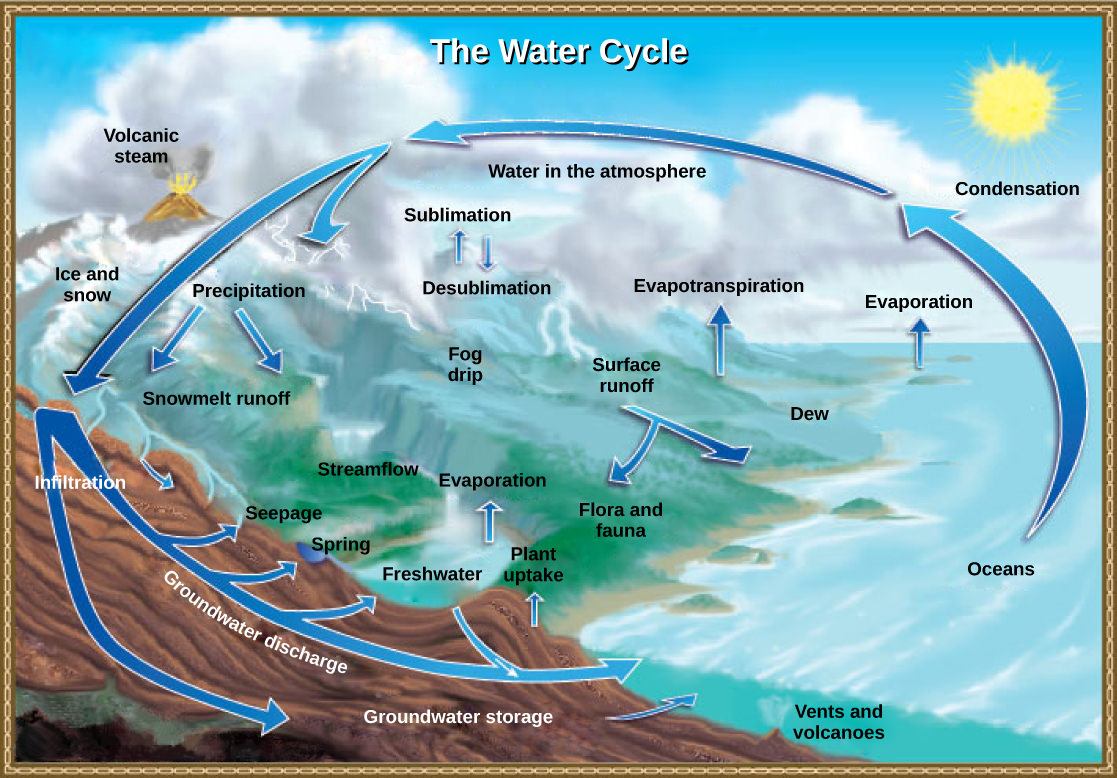
The Water Cycle
The hydrosphere is the area of Earth where water movement and storage occurs: as liquid water on the surface (rivers, lakes, oceans) and beneath the surface (groundwater) or ice (polar ice caps and glaciers), and as water vapor in the atmosphere. The human body is about 60 percent water, and human cells are more than 70 percent water. Of the stores of water on Earth, 97.5 percent is salt water (see Figure 1 below). Of the remaining water, more than 99 percent is groundwater or ice. Thus, less than one percent of freshwater is present in lakes and rivers. Many organisms depend on this small percentage, a lack of which can negatively affect ecosystems. Humans have developed technologies to increase water availability, such as digging wells to harvest groundwater, storing rainwater, and using desalination to obtain drinkable water from the ocean. Although this pursuit of drinkable water has been ongoing throughout human history, freshwater supply remains a major issue. The various processes that occur during the cycling of water are illustrated in Figure 2 below.
The processes include the following:

- evaporation and sublimation
- condensation and precipitation
- subsurface water flow
- surface runoff and snowmelt
- streamflow
The Sun’s energy drives the water cycle as it warms the oceans and other surface waters. This leads to evaporation (liquid water to water vapor) of liquid surface water and sublimation (ice to water vapor) of frozen water, thus moving large amounts of water into the atmosphere as water vapor. Over time, this water vapor condenses into clouds as liquid or frozen droplets, eventually leading to precipitation (rain, snow, hail), returning water to Earth’s surface. Rain reaching Earth’s surface may evaporate again, flow over the surface, or percolate into the ground. Most easily observed is surface runoff: the flow of freshwater over land either from rain or melting ice. Runoff can make its way through streams and lakes to the oceans.
In most natural terrestrial environments, rain encounters vegetation before it reaches the soil surface. A significant percentage of water evaporates immediately from the surfaces of plants. What is left reaches the soil and begins to move down. Surface runoff will occur only if the soil becomes saturated with water in heavy rainfall. Plant roots can take up water in the soil. The plant will use some of this water for its own metabolism, and some of that will find its way into animals that eat the plants. Still, much of it will be lost back to the atmosphere through a process known as transpiration: water enters the vascular system of plants through the roots. It evaporates, or transpires, through the leaves’ stomata (small microscope openings). Ecologists combine transpiration and evaporation into a single term that describes water returned to the atmosphere: evapotranspiration. Water in the soil not taken up by a plant that does not evaporate can percolate into the subsoil and bedrock, forming groundwater.
Groundwater is a significant subsurface reservoir of fresh water. It exists in the pores between particles in dirt, sand, and gravel or in the fissures in rocks. Groundwater can flow slowly through these pores and fissures and eventually finds its way to a stream or lake, where it becomes part of the surface water again. Many streams flow not because they are replenished from rainwater directly but because they receive a constant inflow from the groundwater below. Some groundwater is found very deep in the bedrock and can persist there for millennia. Most groundwater reservoirs, or aquifers, are the source of drinking or irrigation water drawn up through wells. In many cases, these aquifers are being depleted faster than replenished by water percolating down from above.
Rain and surface runoff are major ways minerals, including phosphorus and sulfur, are cycled from land to water. The environmental effects of runoff will be discussed later as these cycles are described.
The Carbon Cycle
Carbon is the second most abundant element in organisms by mass. Carbon is present in all organic molecules (and some molecules that are not organic such as CO2), and its role in the structure of biomolecules is of primary importance. Carbon compounds contain energy, and many of these compounds from dead plants and algae have fossilized over millions of years and are known as fossil fuels. Since the 1800s, the use of fossil fuels has accelerated. Since the beginning of the Industrial Revolution, the demand for Earth’s limited fossil fuel supplies has risen, causing the amount of carbon dioxide in our atmosphere to increase drastically. This increase in carbon dioxide is associated with climate change and is a major environmental concern worldwide.
The carbon cycle is most easily studied as two interconnected subcycles: one dealing with rapid carbon exchange among living organisms and the other dealing with the long-term cycling of carbon through geologic processes. The entire carbon cycle is shown in Figure 3 below.
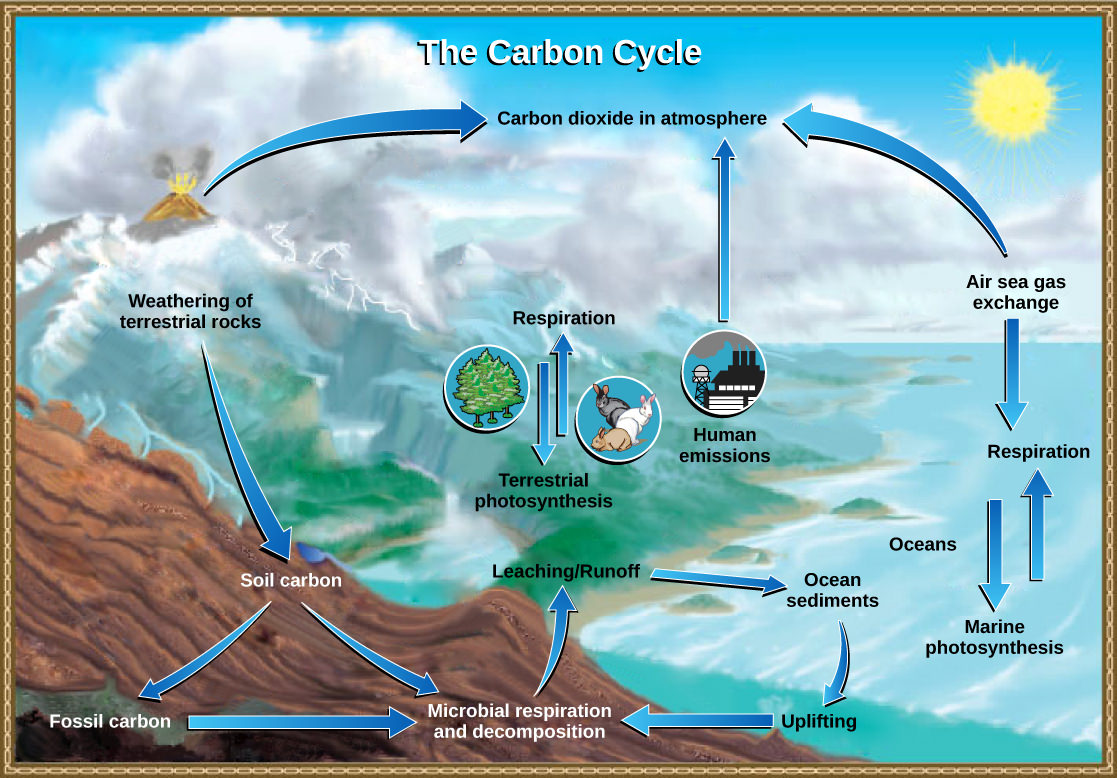
The Biological Carbon Cycle
Organisms are connected in many ways, even among different ecosystems. An example of this connection is the carbon exchange between heterotrophs and autotrophs through atmospheric carbon dioxide. Carbon dioxide (CO2) is the basic building block autotrophs use to build high-energy compounds such as glucose. These organisms use the energy harnessed from the Sun to form the covalent bonds that link carbon atoms together. These chemical bonds store this energy for later use in the process of respiration. Most terrestrial autotrophs obtain their carbon dioxide directly from the atmosphere, while marine autotrophs acquire it in the dissolved form (bicarbonate, HCO3–).
Carbon is passed from producers to higher trophic levels through consumption. For example, when a cow (primary consumer) eats grass (producer), it obtains some of the organic molecules originally made by the plant’s photosynthesis. Those organic compounds can then be passed to higher trophic levels, such as humans, when we eat the cow. At each level, however, organisms perform respiration, a process in which organic molecules are broken down to release energy. As these organic molecules are broken down, carbon is removed from food molecules to form CO2, a gas that enters the atmosphere. Thus, CO2 is a byproduct of respiration. Recall that producers consume CO2 during photosynthesis to make organic molecules. As these molecules are broken down during respiration, the carbon again enters the atmosphere as CO2. Carbon exchange like this potentially connects all organisms on Earth. Think about this: the carbon in your DNA was once part of a plant; millions of years ago, perhaps it was part of a dinosaur.
The Biogeochemical Carbon Cycle
The movement of carbon through land, water, and air is complex, and, in many cases, it occurs much more slowly than the movement between organisms. Carbon is stored for long periods in what are known as carbon reservoirs, which include the atmosphere, bodies of liquid water (mostly oceans), ocean sediment, soil, rocks (including fossil fuels), and Earth’s interior.
As stated, the atmosphere is a major carbon reservoir in the form of carbon dioxide, which is essential to photosynthesis. The level of carbon dioxide in the atmosphere is greatly influenced by the reservoir of carbon in the oceans. The carbon exchange between the atmosphere and water reservoirs influences how much carbon is found in each. Carbon dioxide (CO2) from the atmosphere dissolves in water and reacts with water molecules to form ionic compounds. Some of these ions combine with calcium ions in the seawater to form calcium carbonate (CaCO3), a major component of the shells of marine organisms. These organisms eventually die, and their shells form sediments on the ocean floor. Over geologic time, the calcium carbonate forms limestone, which comprises the largest carbon reservoir on Earth.
On land, carbon is stored in soil as organic carbon due to the decomposition of organisms or from weathering of terrestrial rock and minerals (the world’s soils hold significantly more carbon than the atmosphere, for comparison). Deeper underground are fossil fuels, the anaerobically decomposed remains of plants and algae that lived millions of years ago. Fossil fuels are considered a non-renewable resource because their use far exceeds their formation rate. A non-renewable resource is either regenerated very slowly or not at all. Another way for carbon to enter the atmosphere is from land (including land beneath the ocean’s surface) by the eruption of volcanoes and other geothermal systems. Carbon sediments from the ocean floor are taken deep within Earth by the process of subduction: the movement of one tectonic plate beneath another. Carbon is released as carbon dioxide when a volcano erupts from volcanic hydrothermal vents.
The Nitrogen Cycle
Getting nitrogen into living organisms is difficult. Plants and phytoplankton are not equipped to incorporate nitrogen from the atmosphere (where it exists as tightly bonded, triple covalent N2), even though this molecule comprises approximately 78 percent of the atmosphere. Nitrogen enters the living world through free-living and symbiotic bacteria, which incorporate nitrogen into organic molecules through specialized biochemical processes. Certain species of bacteria can perform nitrogen fixation, the process of converting nitrogen gas into ammonia (NH3), which spontaneously becomes ammonium (NH4+). Bacteria convert ammonium into nitrites (NO2−) and then nitrates (NO3−). At this point, the nitrogen-containing molecules are used by plants and other producers to make organic molecules such as DNA and proteins. This nitrogen is now available to consumers.
Organic nitrogen is especially important to the study of ecosystem dynamics because many ecosystem processes, such as primary production, are limited by the available supply of nitrogen. As shown in Figure 4 below, bacteria eventually convert the nitrogen that enters living systems from organic nitrogen back into nitrogen gas (Figure 4). The process of denitrification is when bacteria convert the nitrates into nitrogen gas, thus allowing it to re-enter the atmosphere.

Human activity can alter the nitrogen cycle by two primary means: the combustion of fossil fuels, which releases different nitrogen oxides, and the use of artificial fertilizers (which contain nitrogen and phosphorus compounds) in agriculture, which are then washed into lakes, streams, and rivers by surface runoff. Atmospheric nitrogen (other than N2) is associated with several effects on Earth’s ecosystems, including the production of acid rain (as nitric acid, HNO3) and greenhouse gas effects (as nitrous oxide, N2O), potentially causing climate change. A major effect of fertilizer runoff is saltwater and freshwater eutrophication, a process whereby nutrient runoff causes the overgrowth of algae, the depletion of oxygen, and the death of aquatic fauna.
In marine ecosystems, nitrogen compounds created by bacteria, or through decomposition, collects in ocean floor sediments. It can then be moved to land in geologic time by uplifting Earth’s crust and thereby incorporated into terrestrial rock. Although the movement of nitrogen from rock directly into living systems has been traditionally seen as insignificant compared with nitrogen fixed in the atmosphere, a recent study showed that this process might indeed be significant and should be included in any study of the global nitrogen cycle.
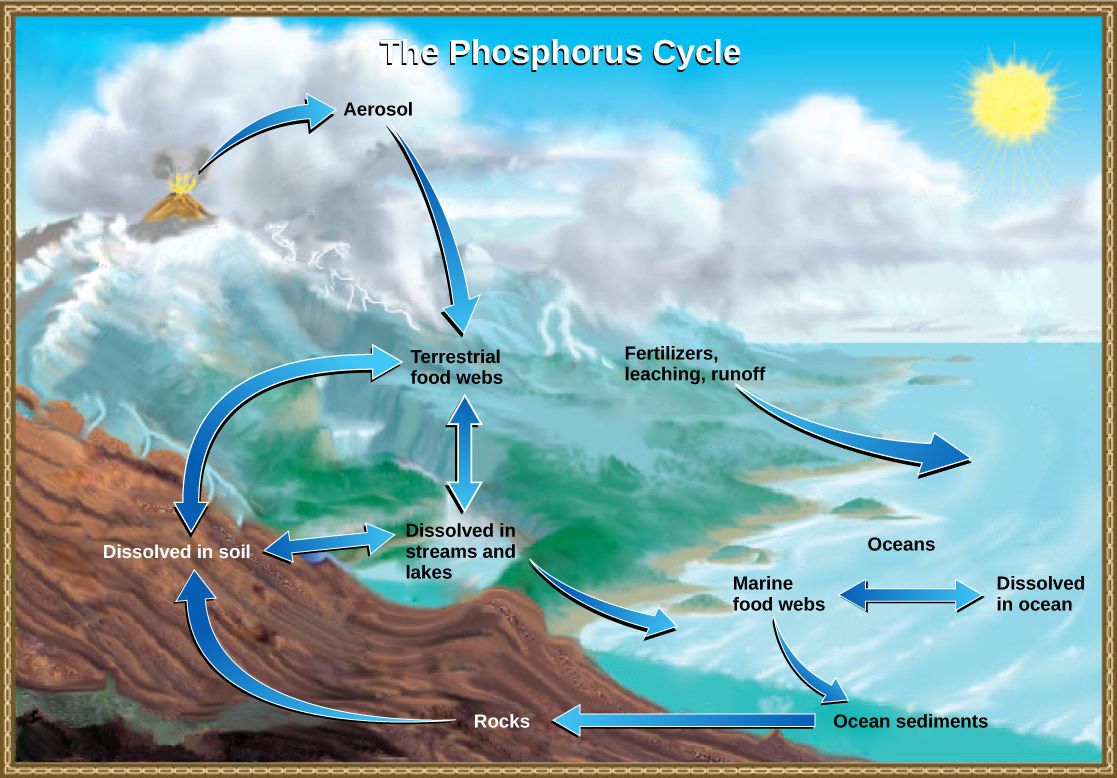
The Phosphorus Cycle
Phosphorus is an essential nutrient for living processes. It is a major component of nucleic acids and phospholipids, and, as calcium phosphate, it makes up the supportive components of our bones. Phosphorus is often the limiting nutrient (necessary for growth) in aquatic, particularly freshwater, ecosystems.
Phosphorus occurs in nature as the phosphate ion (PO43-). In addition to phosphate runoff due to human activity, natural surface runoff occurs when it is leached from phosphate-containing rock by weathering, thus sending phosphates into rivers, lakes, and the ocean. This rock has its origins in the ocean. Phosphate-containing ocean sediments form primarily from the bodies of ocean organisms and from their excretions. However, volcanic ash, aerosols, and mineral dust may also be significant phosphate sources. This sediment then is moved to land over geologic time by the uplifting of Earth’s surface (see Figure 5 below).
Phosphorus is also reciprocally exchanged between phosphate dissolved in the ocean and marine organisms. The movement of phosphate from the ocean to the land and through the soil is extremely slow, with the average phosphate ion having an oceanic residence time between 20,000 and 100,000 years.
Excess phosphorus and nitrogen entering these ecosystems from fertilizer runoff and sewage cause excessive algae growth. The subsequent death and decay of these organisms deplete dissolved oxygen, which leads to the death of aquatic organisms such as shellfish and fish. This process is responsible for dead zones in lakes and at the mouths of many major rivers and for massive fish kills, often during summer (see Figure 6 below).
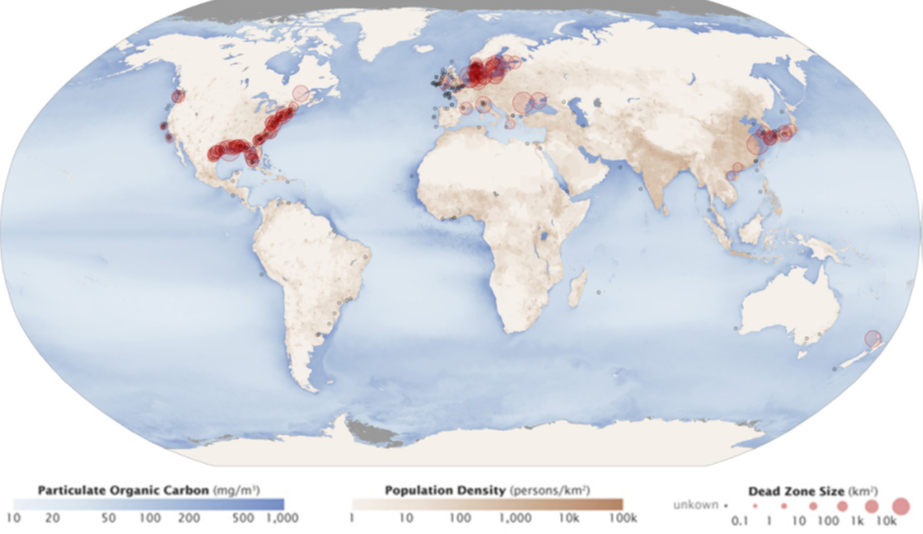
A dead zone is an area in lakes and oceans near the mouths of rivers where large areas are periodically depleted of their normal flora and fauna. These zones are caused by eutrophication coupled with other factors, including oil spills, the dumping of toxic chemicals, and other human activities. The number of dead zones has increased for several years, and more than 400 of these zones were present as of 2008. One of the worst dead zones is off the coast of the United States in the Gulf of Mexico: fertilizer runoff from the Mississippi River basin created a dead zone of over 8,463 square miles. Phosphate and nitrate runoff from fertilizers also negatively affect several lake and bay ecosystems, including the Chesapeake Bay in the eastern United States.
The Sulfur Cycle
Sulfur is an essential element for the molecules of living things. As part of the amino acid cysteine, it is involved in forming proteins. Figure 7 below shows sulfur cycles between the oceans, land, and atmosphere. Atmospheric sulfur is found in the form of sulfur dioxide (SO2), which enters the atmosphere in three ways: first, from the decomposition of organic molecules; second, from volcanic activity and geothermal vents; and third, from the burning of fossil fuels by humans.
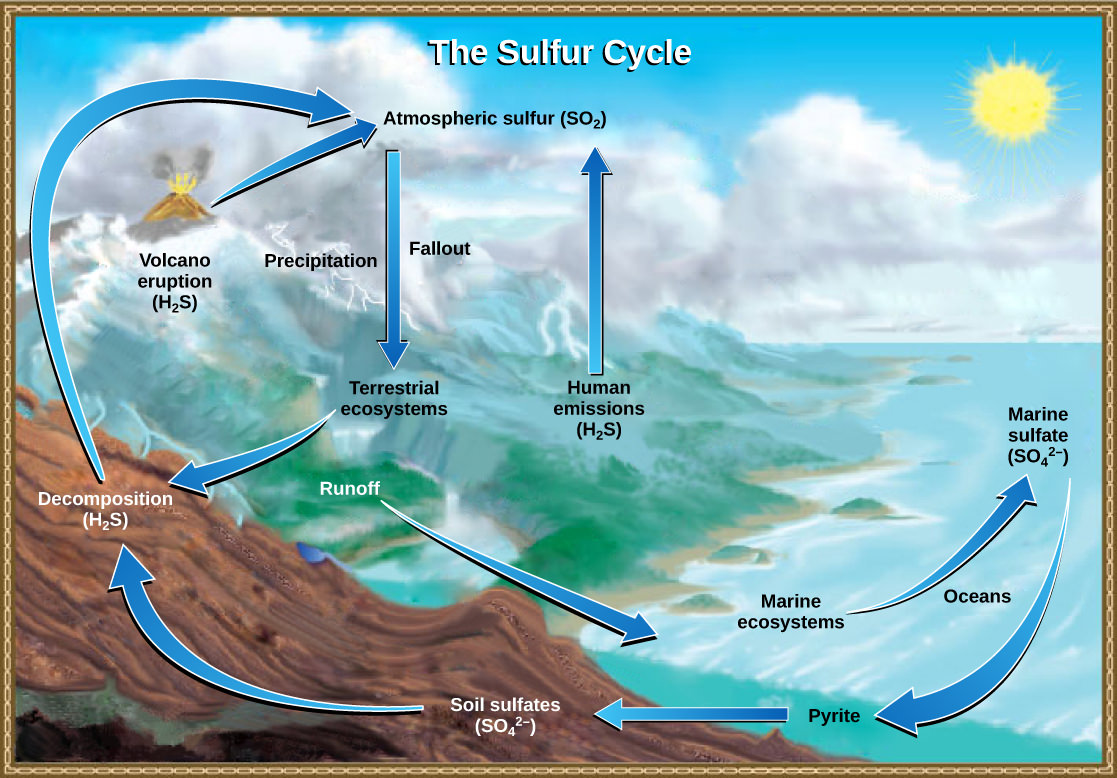
On land, sulfur is deposited in four major ways: precipitation, direct fallout from the atmosphere, rock weathering, and geothermal vents. Atmospheric sulfur is found in the form of sulfur dioxide (SO2), and as rain falls through the atmosphere, sulfur is dissolved in the form of weak sulfuric acid (H2SO4). Sulfur can also fall directly from the atmosphere in a process called fallout. Also, as sulfur-containing rocks weather, sulfur is released into the soil. These rocks originate from ocean sediments that are moved to land by the geologic uplifting of ocean sediments. Terrestrial ecosystems can then use these soil sulfates (SO42-), which enter the food web by being taken up by plant roots. When these plants decompose and die, sulfur is released into the atmosphere as hydrogen sulfide (H2S) gas.
Sulfur enters the ocean in runoff from land, atmospheric fallout, and underwater geothermal vents. Some ecosystems rely on chemoautotrophs using sulfur as a biological energy source. This sulfur then supports marine ecosystems in the form of sulfates.
Human activities have played a major role in altering the balance of the global sulfur cycle. Burning large quantities of fossil fuels, especially coal, releases more hydrogen sulfide gas into the atmosphere. As rain falls through this gas, it creates the phenomenon known as acid rain, which damages the natural environment by lowering the pH of lakes, thus killing many resident plants and animals. Acid rain is corrosive rain caused by rainwater falling to the ground through sulfur dioxide gas, turning it into weak sulfuric acid, which causes damage to aquatic ecosystems. Acid rain also affects the man-made environment through the chemical degradation of buildings. For example, many marble monuments, such as the Lincoln Memorial in Washington, DC, have suffered significant damage from acid rain. These examples show the wide-ranging effects of human activities on our environment and the challenges that remain for our future.
Suggested Supplementary Reading
Bruckner, M. 2018. The Gulf of Mexico Dead Zone. [Website] <https://serc.carleton.edu/microbelife/topics/deadzone/index.html>

